Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 -phase in Al-Mg-Si alloys during aging treatment
-phase in Al-Mg-Si alloys during aging treatmentAhmed, A.*; Uttarasak, K.*; Tsuchiya, Taiki*; Lee, S.*; Nishimura, Katsuhiko*; Nunomura, Norio*; Shimizu, Kazuyuki*; Hirayama, Kyosuke*; Toda, Hiroyuki*; Yamaguchi, Masatake; et al.
Journal of Alloys and Compounds, 988, p.174234_1 - 174234_9, 2024/06
This study aims to clarify the growth process of the -phase in Al-Mg-Si alloys from the point of view of morphology evolution. For this research, the
-phase in Al-Mg-Si alloys from the point of view of morphology evolution. For this research, the  -phase orientation relationship, shape, growth process, misfit value, and interfacial condition between the
-phase orientation relationship, shape, growth process, misfit value, and interfacial condition between the  -phase and Al matrix were investigated using high-resolution transmission electron microscopy (HR-TEM), focus ion beam (FIB), and optical microscope (OM). Results include the identification of {111}
-phase and Al matrix were investigated using high-resolution transmission electron microscopy (HR-TEM), focus ion beam (FIB), and optical microscope (OM). Results include the identification of {111}  facets at the edges of the
facets at the edges of the  -phase, as well as the proposal of two new three-dimensional shapes for the
-phase, as well as the proposal of two new three-dimensional shapes for the  -phase. We purposed the morphology evolution during the growth process of Mg
-phase. We purposed the morphology evolution during the growth process of Mg Si crystal and calculated the misfit to understand the unstable (111)
Si crystal and calculated the misfit to understand the unstable (111) facet has a higher misfit value as compared to the (001)
facet has a higher misfit value as compared to the (001) and (011)
and (011) facets. Our observations provide how they influence the behavior of Mg
facets. Our observations provide how they influence the behavior of Mg Si crystals.
Si crystals.
 neutron diffraction
neutron diffractionZhou, Y.*; Song, W.*; Zhang, F.*; Wu, Y.*; Lei, Z.*; Jiao, M.*; Zhang, X.*; Dong, J.*; Zhang, Y.*; Yang, M.*; et al.
Journal of Alloys and Compounds, 971, p.172635_1 - 172635_7, 2024/01
Times Cited Count:0 Percentile:0(Chemistry, Physical)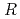 AuGe (
AuGe (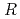 = Y, Gd-Tm, and Lu)
= Y, Gd-Tm, and Lu)Kurumaji, Takashi*; Gen, Masaki*; Kito, Shunsuke*; Ikeuchi, Kazuhiko*; Nakamura, Mitsutaka; Ikeda, Akihiko*; Arima, Takahisa*
Journal of Alloys and Compounds, 947, p.169475_1 - 169475_8, 2023/06
Times Cited Count:1 Percentile:54.26(Chemistry, Physical) Rh
Rh Sn
Sn
Opletal, P.; Duverger-N dellec, E.*; Miliyanchuk, K.*; Malick, S.*; Hossain, Z.*; Custers, J.*
dellec, E.*; Miliyanchuk, K.*; Malick, S.*; Hossain, Z.*; Custers, J.*
Journal of Alloys and Compounds, 927, p.166941_1 - 166941_7, 2022/12
Times Cited Count:2 Percentile:32.54(Chemistry, Physical) CoCrFeNi eutectic high entropy alloy
CoCrFeNi eutectic high entropy alloyYun, D.*; Chae, H.*; Lee, T.*; Lee, D.-H.*; Ryu, H. J.*; Banerjee, R.*; Harjo, S.; Kawasaki, Takuro; Lee, S. Y.*
Journal of Alloys and Compounds, 918, p.165673_1 - 165673_7, 2022/10
Times Cited Count:3 Percentile:32.54(Chemistry, Physical)Zhang, X. X.*; Lutz, A.*; Andr , H.*; Lahres, M.*; Gong, W.; Harjo, S.; Emmelmann, C.*
, H.*; Lahres, M.*; Gong, W.; Harjo, S.; Emmelmann, C.*
Journal of Alloys and Compounds, 898, p.162890_1 - 162890_8, 2022/03
Times Cited Count:6 Percentile:71.08(Chemistry, Physical) Sc
Sc )
) O
O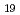 (0
(0 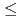 x
x 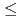 0.2) hexagonal ferrite
0.2) hexagonal ferriteMaruyama, Kenichi*; Tanaka, Seiya*; Kiyanagi, Ryoji; Nakao, Akiko*; Moriyama, Kentaro*; Ishikawa, Yoshihisa*; Amako, Yasushi*; Iiyama, Taku*; Futamura, Ryusuke*; Utsumi, Shigenori*; et al.
Journal of Alloys and Compounds, 892, p.162125_1 - 162125_8, 2022/02
Times Cited Count:2 Percentile:16.86(Chemistry, Physical)Liss, K.-D.*; Harjo, S.; Kawasaki, Takuro; Aizawa, Kazuya; Xu, P. G.
Journal of Alloys and Compounds, 869, p.159232_1 - 159232_9, 2021/07
Times Cited Count:6 Percentile:47.82(Chemistry, Physical)
Yoshii, Kenji; Ikeda, Naoshi*
Journal of Alloys and Compounds, 804, p.364 - 369, 2019/10
Times Cited Count:11 Percentile:54.33(Chemistry, Physical)Dielectric and magnetocaloric measurements are carried out for the chromite TmCrO . This oxide was reported to be multiferroic below the N
. This oxide was reported to be multiferroic below the N el temperature (
el temperature (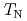 ) of 125 K, likely due to a structural transformation. The dielectric response shows large dielectric constants below 300 K. However, from the analyses of loss tangent, AC conductivity and dielectric modulus, this behavior is rooted in hopping of charge carriers rather than electric dipoles, as proposed for some other chromites. No dielectric anomaly is found at
) of 125 K, likely due to a structural transformation. The dielectric response shows large dielectric constants below 300 K. However, from the analyses of loss tangent, AC conductivity and dielectric modulus, this behavior is rooted in hopping of charge carriers rather than electric dipoles, as proposed for some other chromites. No dielectric anomaly is found at 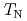 . The magnetocaloric effect shows that the magnetic transitions at
. The magnetocaloric effect shows that the magnetic transitions at 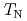 as well as the spin reorientation temperature are of a second order. This result strongly suggests the absence of magnetostructural transition at
as well as the spin reorientation temperature are of a second order. This result strongly suggests the absence of magnetostructural transition at 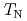 in accord with no observation of ferroelectric transition at this temperature.
in accord with no observation of ferroelectric transition at this temperature.
Iha, Wataru*; Kakihana, Masashi*; Matsuda, Shinya*; Honda, Fuminori*; Haga, Yoshinori; Takeuchi, Tetsuya*; Nakashima, Miho*; Amako, Yasushi*; Gochi, Jun*; Uwatoko, Yoshiya*; et al.
Journal of Alloys and Compounds, 788, p.361 - 366, 2019/06
Times Cited Count:6 Percentile:33.98(Chemistry, Physical)Nishimura, Katsuhiko*; Matsuda, Kenji*; Lee, S.*; Nunomura, Norio*; Shimano, Tomoki*; Bendo, A.*; Watanabe, Katsumi*; Tsuchiya, Taiki*; Namiki, Takahiro*; Toda, Hiroyuki*; et al.
Journal of Alloys and Compounds, 774, p.405 - 409, 2019/02
Times Cited Count:3 Percentile:17.96(Chemistry, Physical)Moro, Takuya*; Kim, J.*; Yamanaka, Satoru*; Murayama, Ichiro*; Kato, Takanori*; Nakayama, Tadachika*; Takeda, Masatoshi*; Yamada, Noboru*; Nishihata, Yasuo; Fukuda, Tatsuo; et al.
Journal of Alloys and Compounds, 768, p.22 - 27, 2018/11
Times Cited Count:17 Percentile:66.2(Chemistry, Physical)Hosokawa, Shinya*; Kimura, Koji*; Yamasaki, Michiaki*; Kawamura, Yoshihito*; Yoshida, Koji*; Inui, Masanori*; Tsutsui, Satoshi*; Baron, A. Q. R.*; Kawakita, Yukinobu; Ito, Shinichi*
Journal of Alloys and Compounds, 695, p.426 - 432, 2017/02
Times Cited Count:3 Percentile:16.72(Chemistry, Physical) 1
1 -type AlNi
-type AlNi alloy at high pressure and temperature
alloy at high pressure and temperatureEndo, Naruki*; Saito, Hiroyuki; Machida, Akihiko; Katayama, Yoshinori
Journal of Alloys and Compounds, 645(Suppl.1), p.S61 - S63, 2015/10
Times Cited Count:0 Percentile:0.01(Chemistry, Physical)Kim, Jae-Hwan; Nakamichi, Masaru
Journal of Alloys and Compounds, 640, p.285 - 289, 2015/08
Times Cited Count:3 Percentile:17.9(Chemistry, Physical)Kim, Jae-Hwan; Nakamichi, Masaru
Journal of Alloys and Compounds, 638, p.277 - 281, 2015/07
Times Cited Count:5 Percentile:27.56(Chemistry, Physical)Kim, Jae-Hwan; Nakamichi, Masaru
Journal of Alloys and Compounds, 585, p.63 - 68, 2014/02
Times Cited Count:14 Percentile:55.58(Chemistry, Physical)Abe, Hiroshi; Aone, Shigeo*; Morimoto, Ryo*; Uchida, Hirohisa*
Journal of Alloys and Compounds, 580(Suppl.1), p.S219 - S221, 2013/12
Times Cited Count:5 Percentile:31.8(Chemistry, Physical)no abstracts in English
 Fe M
Fe M ssbauer spectroscopy of RFe
ssbauer spectroscopy of RFe (R = Y, Gd) hydrides synthesized under ultra-high-pressure hydrogen
(R = Y, Gd) hydrides synthesized under ultra-high-pressure hydrogenMitsui, Takaya; Masuda, Ryo*; Seto, Makoto; Hirao, Naohisa*; Matsuoka, Takehiro*; Nakamura, Yumiko*; Sakaki, Koji*; Enoki, Hirotoshi*
Journal of Alloys and Compounds, 580(Suppl.1), p.S264 - S267, 2013/12
Times Cited Count:8 Percentile:42.73(Chemistry, Physical)Chen, Y.*; Feng, X.*; Kasukabe, Yoshitaka*; Yamamoto, Shunya; Yoshikawa, Masahito; Fujino, Yutaka*
Journal of Alloys and Compounds, 577(Suppl.1), p.S18 - S24, 2013/11
Times Cited Count:2 Percentile:17(Chemistry, Physical)Epitaxial transformation processes of titanium films due to Nitrogen-implantation have been clarified through in-situ observations by using transmission electron microscope(TEM)and electron energy loss spectroscope, along with molecular orbital calculations. The N
 ions with 62 keV are implanted into as-deposited Ti films which consist of hcp-Ti and TiH
ions with 62 keV are implanted into as-deposited Ti films which consist of hcp-Ti and TiH with preferred orientations, in the 400 kV analytic high resolution TEM combined with ion accelerators at JAEA Takasaki. Thus, titanium nitride (TiN
with preferred orientations, in the 400 kV analytic high resolution TEM combined with ion accelerators at JAEA Takasaki. Thus, titanium nitride (TiN ) films with preferred orientations are epitaxially formed by the inheritance of partial atomic arrangement of hcp-Ti or TiH
) films with preferred orientations are epitaxially formed by the inheritance of partial atomic arrangement of hcp-Ti or TiH in as-deposited Ti films and by the occupation of octahedral sites by N atoms, which elucidates that epitaxial transformation of hcp-fcc Ti sublattices occurs.
in as-deposited Ti films and by the occupation of octahedral sites by N atoms, which elucidates that epitaxial transformation of hcp-fcc Ti sublattices occurs.